Reviewed and Approved by Kaci Foote, Accreditation Manager, FSNS Certification & Audit

1-Minute Summary
- Safe Feed/Safe Food Version 8 is intended to clarify requirements, eliminate redundancy, and define written procedure expectations more clearly.
- Overall certification requirements are generally the same, but the structure has changed.
- Version 8 includes 12 main categories with 62 clauses, plus a separate category and clause for the Safe Feed/Safe Food seal.
- Version 7.0 had 29 mandatory clauses. Version 8 replaces them with 26 core requirements that carry higher nonconformance scores.
- Facility ratings such as Excellent, Good, and Compliant have been removed, but audit scores still affect certification cycles.
- Minor and major audit findings now have a standardized response time of 40 business days but can depend upon the Certification Body’s internal requirements.
- A new Safe Feed/Safe Food Certified Warehouse, Storage and Distribution certification option is available for non-manufacturing operations.
What Changed in Safe Feed/Safe Food Version 8?
Safe Feed/Safe Food Version 8 has been released, with implementation expected by June 1, 2026. Facilities with Safe Feed/Safe Food certification – or those preparing for certification – should review the updated requirements before their next audit.
First, take a sigh of relief – overall certification requirements remain the same in Version 8. However, the updated standard does reorganize and clarify the program so facilities can better understand what must be documented, implemented, audited, and maintained.
Our FSNS Certification & Audit team breaks down the key changes below. FSNS C&A is a Safe Feed/Safe Food Certifying Body, so contact us to schedule your audit.
CTA Button: [Schedule Your Safe Feed/Safe Food Audit Today]
Safe Feed/Safe Food Version 8 At a Glance
| Version 7 | Version 8 |
| 83 clauses | 62 clauses |
| 29 mandatory clauses | 26 core requirements |
| Ratings assigned | Ratings removed |
| Minor/major response times varied | 40 business days |
| Guidance included in requirement text | Guidance moved to Appendix B |
Guidance Information Moved to Appendix B
Version 8 separates guidance from the requirements text. Guidance information has been removed from the requirements and placed in Appendix B of the Safe Feed/Safe Food Certified Facility Guidance Document. This should make the auditable requirements easier for facilities and auditors to identify.
Key Safe Feed/Safe Food Version 8 Changes – FSC36 Removed
Version 8 now refers to the business seeking certification as the “facility” instead of the “supplier.” It also replaces the term “vendor” with “supplier.” Facilities should update internal documents, supplier packets, audit preparation materials, and customer-facing references as needed.
The update also removes references to FSC36. FSC codes are assigned to SQFI certifications, and Safe Feed/Safe Food is not an SQFI certification. AFIA continues to work closely with SQFI on the registration and certification processes.
Safe Feed/Safe Food Certification Version 8 Change Map
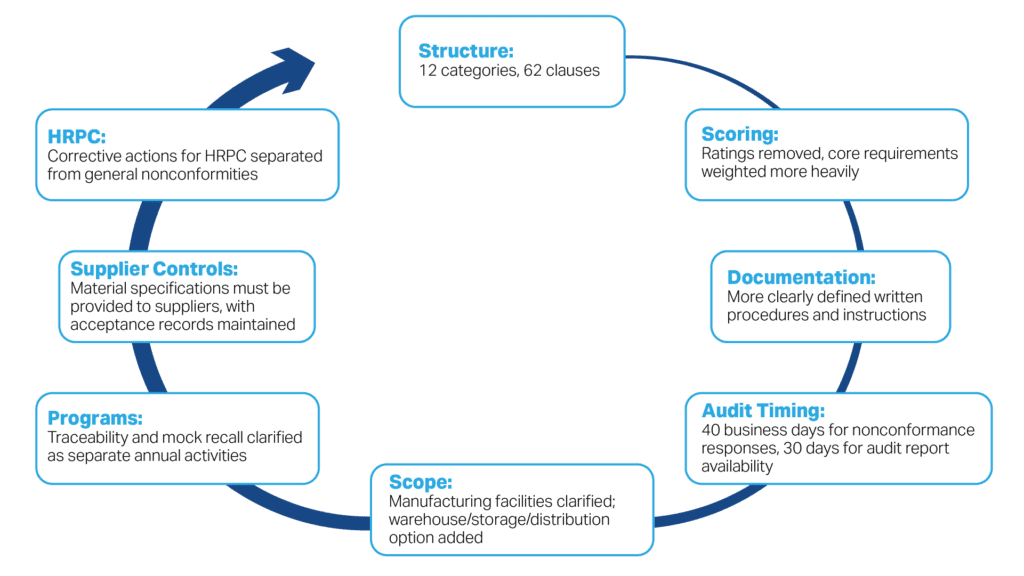
Requirements are Reorganized and Streamlined
Version 7.0 included 83 clauses. Version 8 includes 12 main categories with 62 clauses, plus a separate category and clause for the Safe Feed/Safe Food seal. The intent is to clarify the requirements and eliminate redundancy.
The 12 main Version 8 categories are:
- Management Commitment
- Quality and Animal Food Safety System
- Personnel
- Facility Operations
- Finished Product Realization
- Infrastructure and Maintenance
- Pest Management and Control
- Verification Activities
- Internal Audits
- Suppliers for Materials and Services
- Animal Food Safety Programs and Requirements
- Requirements for a Hazard Requiring a Preventive Control
This structure should make the requirements easier to navigate, but sites should update internal audit checklists and gap assessments to match the new organization.
Safe Feed/Safe Food Version 8 Core Requirements Replace Mandatory Clauses
Version 7.0 had 29 mandatory clauses. Version 8 replaces them with 26 clauses identified as core requirements.
Core requirements carry higher nonconformance scores than other clauses:
- Minor nonconformance: 3 points for core requirements vs. 1 point for other clauses.
- Major nonconformance: 10 points for core requirements vs. 7 points for other clauses.
Certified facilities should identify each core requirement and confirm that supporting evidence is ready, including current procedures, complete records, assigned responsibilities, training evidence, and corrective action documentation where applicable.
Rating Scores No Longer Assigned in Safe Feed/Safe Food Version 8
Version 8 removes facility ratings such as Excellent, Good, and Compliant. SF/SF explains that ratings could discourage facilities and auditors from reporting nonconformities, even though findings should help facilities drive continuous improvement.
Audit scores still matter. Facilities that score above 85 are compliant for two years. Facilities that score 70 to 85 are compliant for one year.
Surveillance Audits Are Pass/Fail
Surveillance audits are not scored and are recorded as Pass/Fail. During the surveillance audit year, the facility maintains the score obtained during the last on-site audit. Facilities should confirm surveillance audit expectations with their certification body when planning their audit cycle.
Corrective Action Timelines Have Changed
Version 8 standardizes the response time for minor and major audit findings to 40 business days.
Certification bodies must make the audit report available to the facility within 30 days from the last day of the audit. However, this also depends on the Certification Body’s internal requirements.
These changes are intended to give facilities and certification bodies more time to report and respond effectively. Sites should update corrective action procedures, internal tracking tools, and certification planning timelines.
Audit Report Timing Has Changed
Version 7 required the certification body to provide the audit report within 14 calendar days from the last day of the audit.
Safe Feed/Safe Food Version 8 changes this to 15 business days.
Sites should account for this timing when planning customer updates, certification renewals, and management review activities.
Safe Feed/Safe Food Requires More Written Procedures
Safe Food/Safe Feed Version 8 more clearly defines when written documents, policies, or procedures are required. At least 20 clauses require written documentation if applicable to the facility.
For many facilities, this may be the most important preparation step. If a process is being followed but is not clearly documented, Version 8 may create an audit gap.
Manufacturing and Warehouse Certification Paths Are Clearer
The guidance more clearly states that Safe Feed/Safe Food Certified Facility certification applies to manufacturing facilities, not warehouses.
Facilities may mark some clauses as not applicable when appropriate. However, if more than 18 clauses, or 30%, are listed as not applicable, the facility is not eligible for certification. Certification bodies are responsible for ensuring the number of N/A clauses does not exceed 18.
Version 8 also introduces a Safe Feed/Safe Food Certified Warehouse, Storage and Distribution certification option. Warehouses, storage facilities, and distribution facilities may use the SFSFv8 program guidance document with the Warehouse, Storage and Distribution audit form, which uses fewer clauses and provides a different certification seal.
Traceability and Mock Recall Requirements Are Separate
Version 8 clarifies that product traceability is different from a mock recall. A traceability exercise is required annually. A mock recall is also required annually to assess the recall procedure and its effectiveness.
Supplier Specification Requirements Are Clearer
Specifications for materials used in manufacturing must be provided to suppliers. Facilities must maintain a record of the supplier’s acceptance or approval. A facility may use the supplier’s specifications if it maintains a record noting its acceptance.
HRPC Corrective Actions Are Separated
Version 8 separates corrective actions for audit nonconformities from corrective actions for hazards requiring a preventive control. Facilities should review their animal food safety plan, hazard analysis, preventive control records, and corrective action procedures to ensure the difference is clear.
The QFS Leader Role Is More Central
Safe Feed/Safe Food Version 8 replaces the previous standalone PCQI clause with a requirement for the Quality and Food Safety Leader. The QFS Leader must still be a Preventive Controls Qualified Individual.
Facilities should confirm that the QFS Leader is identified, trained, qualified, and involved in the animal food safety system. Our FSPCA Preventive Controls for Animal Food Course provides FDA-recognized PCQI animal food training.

How Certified Facilities Should Prepare for Their Next Safe Feed/Safe Food Audit
Sites with Safe Feed/Safe Food certification should focus on a few practical actions before the next audit:
- Confirm whether your site is eligible as a manufacturing facility or should pursue warehouse/storage/distribution certification.
- Update internal audit tools to match Version 8 categories and clause numbers.
- Identify all 26 core requirements.
- Review written procedures and instructions.
- Confirm QFS Leader and PCQI documentation.
- Separate traceability exercise records from mock recall records.
- Review supplier specification and acceptance records.
- Update corrective action procedures to reflect the 40-business-day timeline.
- Plan audit scheduling, report timing, and recertification milestones.
The Bottom Line on Safe Feed/Safe Food Certification Under Version 8
Safe Feed/Safe Food Version 8 is primarily a clarification and documentation-focused update. The requirements are generally the same, but the structure, terminology, written procedure expectations, scoring approach, and certification options have changed.
For certified facilities, the priority is to understand the new structure, prepare for core requirement scoring, update written procedures, and confirm that the quality and animal food safety system is ready before the next audit.