New foods introduce fresh flavors, new benefits, and new customers. But novel formulations also raise safety questions that need answers long before a product reaches the shelf. Aaron Pleitner, PhD, Director of Microbiology Technical Services at Certified Group, shares key considerations for evaluating the safety of emerging ingredients and new food concepts.
What are the first food safety issues innovators should consider when developing a new or unconventional formulation?
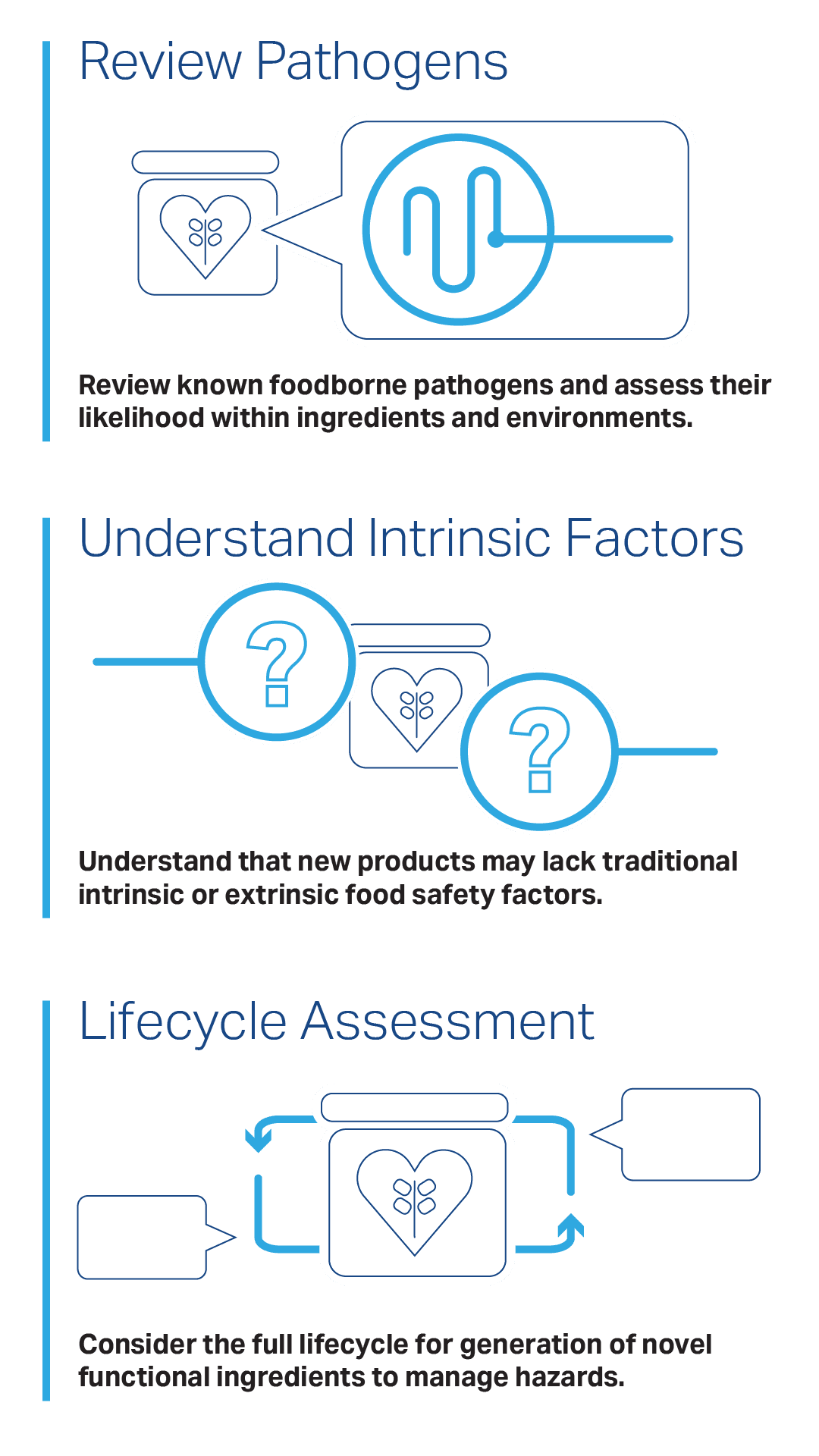
Take it back to the basics! Start with reviewing the known foodborne pathogens and assess their likelihood for prevalence within ingredients and environments pertaining to the new or unconventional formulation.
Understand that you may be developing a new product that no longer uses the longstanding intrinsic or extrinsic factors in more traditional products.
Further, with novel functional ingredients and products, the full lifecycle for generation of those ingredients should be taken into consideration to facilitate an innovator employing “smart design” to manage that hazard.
Novel Foods Safety Considerations
How can formulators identify the right microbial hazards for ingredients with limited data, such as emerging botanicals?
Discuss and partner with your ingredient suppliers!
Increasingly, the industry is relying upon supply chain preventive controls to provide multiple hurdles of management for microbiological risk. In understanding the process that goes into making the ingredient as well as the control applied, a more educated approach can be taken to identifying potential hazards of control.
Further, a holistic approach should be taken for both overall microbiological presence as well as targeted pathogens of concern in the ingredients. Hygiene indicators and background microflora testing isn’t just for environmental samples. Collecting historical data around Total Plate Count (TPC), Enterobacteriaceae, and Aerobic/Anaerobic Sporeformer testing across multiple production batches from different seasons and geographical regions can provide insight into how abundant the microbiological flora is in the ingredient. This helps define data-driven food microbiology criteria for ingredients/product and will facilitate a right fit for routine microbiology testing and tolerance limits while helping to predict prevalence and abundance with upper limits from batch to batch.
Lastly, either employ the expertise in-house or leverage existing databases for thorough recall and outbreak history for similar ingredients, including botanicals, as it pertains to sourcing and production, in order to approach things in an educated fashion.
When is routine testing not enough, and when should teams plan for custom studies like challenge tests or thermal validation?
Overall, routine testing is only a verification of controls and assurance that the finished product design can tolerate the microbiological populations. To define the robustness for the product formulation in-market, a challenge study should be conducted for novel formulations to, most importantly, protect the consumer as well as the brand reputation of the company.
Further, a food safety expert and process authority should be leveraged to determine if a thermal process is validated for the formulation in question or if additional process validation work is needed to have confidence in the critical control points in a food safety program.
How useful is comparing a new ingredient to similar foods when assessing potential hazards and safety expectations?
It can be a great start toward proper risk assessment. Leveraging existing data on like-for-like products facilitates faster and more accurate identification of potential hazards and an understanding of the robustness of interventions.
Having said that, it is critical to partner or employ in-house a seasoned microbiologist who can perform risk assessments and has the wherewithal to apply data from similar products/ingredients to the new ingredient (if possible).
Additionally, applying existing data to new ingredients can be possible and is common; however, verification through testing and monitoring programs is a key pillar to include in programs to constantly confirm these assumptions.
“Leveraging existing data on like-for-like products facilitates faster and more accurate identification of potential hazards and an understanding of the robustness of interventions.”
– Aaron Pleitner, PhD | Director of Microbiology Technical Services, Certified Group
How does understanding the production process, including hot holding and pH shifts, help determine the correct kill step or validation needs?
It is paramount to have a thorough understanding. All intrinsic characteristics of a formulation can have an impact on the adequacy of kill steps.
Specifically, pH; moisture content; starting temperature; and macronutrients of fat, carbohydrates, and protein can all have an impact on both thermal conductivity of the food material as well as bacterial susceptibility or tolerance to mechanisms of inactivation, like thermal treatment. It is critical to assess each of these factors individually as well as synergistically to determine if validation is needed for a new or updated formulation.
Why do plant‑based or high‑fat beverages require different time and temperature targets for microbial reduction than dairy products?
Plant-based matrices may need adjusted time and temperature targets when compared to dairy products. The reason for this is both microbiological as well as pertaining to quality and sensory.
Starting with quality, the viscosity and thermal conductivity of an almond paste in solution is going to exhibit different behaviors physically when pumping through production lines than a fluid milk that has gone through a homogenizer. With varying viscosities, resultant flow rates and thermal conductivity throughout the material, the time and temperature for either a plate heat exchanger or tube-in-tube process are going to vary to prevent scorching or unwanted caramelization of sugars.
Secondly, speaking specifically to microbiological behavior, differing salts and sugar content, available moisture and fat contents and types (i.e. saturated vs unsaturated) are going to provide either protective insulation against high temperature or provide increased sensitivity based on these factors.
A very clear example of this is the tolerance that Salmonella has exhibited for high temperature in low moisture products compared with a high moisture food matrix. High sugar, low moisture, and high fat matrices are all facilitating similar benefits toward microbiological populations whereupon the limitation of unbound water results in bacterial resilience towards thermal processes.
How should companies factor real consumer behavior into safety evaluations, especially for products left unrefrigerated or stored after opening?
We live in a fast-paced world where convenience is king. In addition to this, however, consumers are also demanding nutritious options along with this convenience.
Novel formulation is at this intersection of fulfilling consumer expectations. Primary packaging and product artwork is the place to provide succinct yet detailed instructions to the customer, ensuring that at the time of consumption, the product meets both the intended quality and safety.
Advertising and product packaging are the primary vehicles to educate the consumer and bring awareness on how to handle products. There is a reason why package instructions require rigorous review. Product handling and heat/re-heating need to be prescriptive and clearly visible – but not onerous.
This is where an in-depth understanding of how a product formulation will hold up during instances of temperature abuse is crucial as part of product design. Instruction to the consumer for refrigeration after opening or after hydration will facilitate decreasing the likelihood of product decline.
Also, an understanding of the manufacturing site that is producing the product at time of commerciliazation and scaling is paramount to apply either re-heat or cook instructions on the label (based on if the site is an RTE, RTC, or raw facility).
All in all, cross-collaboration and partnership with product formulators, food safety experts, and operations is critical to adequately design, scale, and produce novel products. Strong partnerships protect product quality and safety by pairing microbiological risk assessment expertise with verification testing and monitoring across ingredients, formulation, unit operations, and on-shelf conditions.
Talk to our team about designing a shelf life study, challenge study, process validation, or other contract research project to help ensure the safety and quality of your novel foods!